We are at an inflection point: Have HTA and PBS reform in Australia been a failure?
- Brendan Shaw
- Mar 31
- 10 min read
Updated: Apr 1
Brendan Shaw

"The government would like to achieve further PBS savings by making greater use of generic drugs to allow headroom for the expensive, but cost-effective, innovator drugs that are likely to be subsidised by the PBS in the near future.”
- The Hon Tony Abbott MP, Minister for Health and Ageing, 17 May 2006, Sydney Morning Herald
“In challenging economic times, both parties have a mutual interest in ensuring that PBS expenditure remains sustainable and the PBS remains responsive to change and to the availability of new and innovative medicines.”
- The Hon Nicola Roxon MP, Minister for Health and Ageing, May 2010, Letter to Medicines Australia for MoU.
“This reform package is designed to be a sensible start that focuses on longer-term structural reform to enable ongoing investment in new medicines while ensuring they remain affordable for patients and taxpayers.”
- The Hon Sussan Ley MP, Minister for Health and Ageing, 27 May 2015, Sussan Ley News
“These agreements will also oversee improvements to Australia’s world-class medicines assessment processes to ensure Australia keeps pace with the ever-changing advances in medicine and so that Australians can access the latest health technologies as soon as possible.”
- The Hon Greg Hunt MP, Minister for Health and Aged Care, 7 Sep 2021, Media Release
I was invited to speak this week at a Monash University forum on the topic of how health technology assessment (HTA) policy and processes have evolved in Australia in the face of challenges and how successful this has been.
To be honest, as one of the people who lead the Australian pharmaceutical industry and contributed to the reform process on and off through the last 20 years, I found myself confronted by the reality that I may have failed.
While preparing the speaking points and slides for my talk, reflecting on more than 20 years as a player, commentator, teacher and occasional whinger about HTA and Pharmaceutical Benefits Scheme (PBS) reform in Australia, I realised that many of the promises of reform to HTA had yet to be delivered.
It was a sobering moment.
More that 20 years after the pricing reform program for Australia's PBS started, the emerging question is whether the entire reform program for HTA and the PBS has been worth it.
While, yes, today the PBS is financially sustainable and net expenditure is actually falling, the reforms have to date failed to improve and speed up Australians' access to new and innovative medicines. Money from savings derived from pharmaceutical companies has not been sufficiently re-invested in new medicines, and Australia's performance compared to other countries in funding new medicines has not improved.
If anything, Australia's competitiveness as a market for medicines has worsened.
This is despite all the excellent collaborative work the companies, Medicines Australia, patient groups and clinicians have done over the years to provide solutions to government.
Over the same period, other parts of government spending have grown. Be it defence spending, the National Disability Insurance Scheme, AUKUS submarines, or fuel tax rebates for mining companies, all of these expenditure areas have kept pace with growth in Australia's economy. But not the PBS. This has largely been due to savings paid for by pharmaceutical companies.
What was PBS reform for?
The reform of pricing policies for Australia's PBS first started with the introduction of the first statutory 12.5% price reduction for generic medicines. But the momentum for policy reform in the PBS really started with the split of the Scheme into two formularies: F1 (single brand medicines) and F2 (multiple brand medicines) in 2007. Combined with the introduction of price disclosure to generate savings for government from market competition in the F2 medicines market, and statutory price cuts for both F1 and F2 medicines, these reforms were designed to free up funding in the PBS to prepare it for the 21st century.
The critical point is to understand the policy logic underpinning the reform program.
From the start more than 20 years ago, there have been two dual policy objectives of PBS reform:
Improve the financial sustainability of the PBS by generating savings from price reductions, and
Using the financial headroom provided by those savings to improve the way Australians have access to new medicines and, in more recent times, secure supply of generic medicines.
Over the years, various government health ministers have explained that PBS reform is about both ensuring the financial sustainability of the PBS and about improving Australians' access to new medicines.
The problem is that after two decades of policy changes these two policy objectives have not been equally pursued. While PBS savings measures and price cuts have generated rivers of gold for federal government coffers, the progress in implementing HTA reforms has been nowhere near as successful.

PBS savings
The short story is that since the beginning of the century, the government's spending on the PBS has fallen as a share of Australia's national income.


The combined effect of a range of price cuts for both F1 and F2 medicines, an increasing emphasis on budget management in the government's handling of new medicines listing, and a growing use of net pricing and budget caps has kept a lid on PBS expenditure growth.
As a nation, we are spending less of our national income on the PBS than we were at the beginning of the century.
Pharmaceutical companies today pay back around 35% of the cost of the PBS to government each year.
Could you imagine going to all the service providers to the National Disability Insurance Scheme around Australia and asking them to regularly pay back 35% of their revenue each year to government? Imagine what the reaction would be.


Rebate payments by pharmaceutical companies back to the government for the PBS come from two sources: confidential net pricing agreements and budget caps on expenditure for individual medicines. These rebates have been growing each year such that today the amount of money the innovative pharmaceutical companies pay in rebates to government almost equals the amount of net revenue they receive from F1 and single brand combination medicines after rebates on the PBS.
Think about that for a second. The innovative pharmaceutical companies each year pay almost as much back to the government as they earn in total in F1 and single brand combination products. Once you take pharmacy and wholesaler distribution costs out of that, it's likely that the innovative industry already pays equal to, or more, money in rebates to the government than it receives in selling innovative medicines through the PBS in Australia.
One has to ask a fundamental question: Is this commercially sustainable from an industry-wide perspective over the longer term?
Over the long-term, can an industry continue to earn less net revenue than it pays back in repayments back to government and expect to continue prioritising Australia as an important market for new and innovative medicines?
We know that governments over the last two decades have not re-invested those rebates and the savings back into the PBS. For example, the Department of Health, Disability and Ageing has conceded that not all of the $28 billion saved through F2 price disclosure since 2007 was reinvested into the PBS. Moreover, the tight pricing controls and budget caps imposed by government on PBS listings of new medicines show up as rebate payments in the Budget each year. At the same time, the list of new medicines that have received a positive recommendation from the Pharmaceutical Benefits Advisory Committee (PBAC) but have not proceeded to listing seems to be growing.
This is not what the original PBS reform agenda was about.

HTA reform?
It's fair to say that there have been reforms to the HTA processes and administration of the PBS since the beginning of the century. Lots of them. We've seen the introduction of cost recovery of PBS listing processes, improvements to transparency, accountability and stakeholder participation in the PBAC processes resulting from the Australia-United States Free Trade Agreement, some improvements to processes through streamlining and allowing partial parallel application processes for TGA and PBAC evaluations, and some efforts to improve the efficiency of the administration around pricing and HTA negotiations.
But these have all been process and administration. There has been less progress making substantive changes to the assumptions, methods or valuations used in valuing and pricing medicines.
In many cases, Australia is still using the same valuations, assumptions and methods in its HTA evaluations that have been used since the 1980s and early 1990s. Whether it's cost-effectiveness thresholds that are not indexed to inflation, 5% discount rates that were decided in the 1980s, arguments about choice of comparator that have gone on for decades, or the lack of recognition in HTA evaluations of broader social and economy-wide productivity benefits, there is a laundry list of reforms to out-of-date methods and valuations that have not progressed.

These and other outdated valuation models in the PBS listing system end up manifesting in more stringent net pricing arrangements and budget caps for companies that they then pay back in rebates. This trend risks making the supply of new medicines on the PBS commercially unviable. It's why we're hearing more noise about medicines not proceeding to listing, why companies keep coming back to the PBAC for pricing reviews, and why there's more talk in the industry about private market options in Australia.
There is a large report, the Health Technology Assessment Policy and Methods Review – Final report, which has been sitting with the government for almost two years while it considers a report back from another advisory committee. While there are good options for reform in the HTA Review Report, it is not perfect. Some of the Report's recommendations lead to more reviews, discussions and committees, rather than definitive policy action. Many of the Report's actual recommendations don't go far enough, or rehash old ideas from 20 years ago - an indication of how slow progress on HTA reform has been in Australia. (You can read my earlier review of the report here).
But the time for delaying progress on HTA reform is over. The industry has been patient, working constructively with governments for two decades on PBS reform, but the time to deliver on improvements for the benefit of Australian patients and Australia's productivity performance has arrived.
Despite more than 20 years of savings being delivered to the PBS, Australia still hasn't improved its ranking in international comparisons of patients' access to new medicines. Australians still wait too long for new therapies compared to their counterparts in other countries, and we often don't get the range of innovative medicines that people in the US, Europe, Japan and other peers get access to.
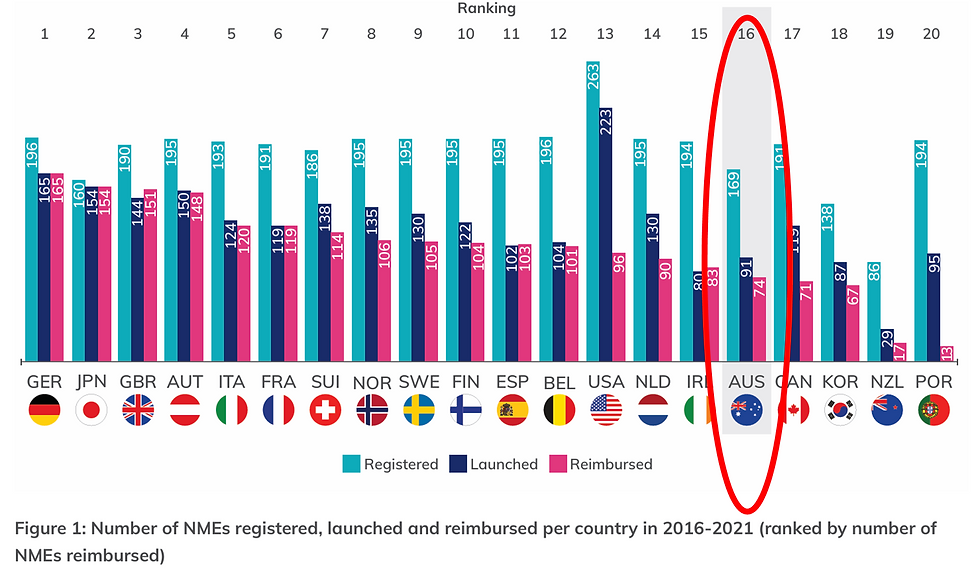
Source: Medicines Australia. 2022. Medicines Matter 2022, p. 6, https://www.medicinesaustralia.com.au/wp-content/uploads/sites/65/2023/04/Medicines-Matter-2022-FINAL.pdf, accessed 17/3/2026.

Source: PhRMA. 2023. Global Access to Medicines Report, p. 28, https://cdn.aglty.io/phrma/global/resources/import/pdfs/2023-04-20%20PhRMA%20Global%20Access%20to%20New%20Medicines%20Report%20FINAL-1.pdf, accessed 17/3/2026.

Source: PhRMA. 2023. Global Access to Medicines Report, p. 17, https://cdn.aglty.io/phrma/global/resources/import/pdfs/2023-04-20%20PhRMA%20Global%20Access%20to%20New%20Medicines%20Report%20FINAL-1.pdf, accessed 17/3/2026.
Australia's relative importance in the global medicines market for medicines is also falling. Measured as a ratio of domestic sales to US sales, Australia's rank in market size has fallen over time, meaning that companies making commercial decisions about the number and timing of new medicines listings may look upon Australia as a less important market.
Global top 20 countries ranking and invoice spending relative to the United States, rank and share of US invoice spending (%)

Source: IQVIA Institute for Human Data Science, Global Use of Medicines Outlook, various years, https://www.iqvia.com/insights/the-iqvia-institute
For decades, I have heard politicians, bureaucrats, PBAC members and academics say that Australia can't afford to fund more new medicines on the PBS for Australian patients. That the government's Budget can't handle it, that there are 'opportunity costs', that we need to restrict PBS spending to allow other important areas of government spending, and that we can't afford to let the PBS have excessive expenditure growth.
This, at a time when real PBS spending is declining, our PBS-to-GDP ratio has been deteriorating since the PBS reform agenda started, and the PBS has been used to fund growth in other spending areas that do not have the same scrutiny and discipline as the PBS. In addition, more and more signals in the data and in comments in the industry suggest that Australia is losing in the race to make new medicines available to Australians.
To cap it off, the current Medicines Australia - Australian Government Strategic Agreement, an Agreement that enjoys bipartisan political support, has an explicitly stated policy goal of maintaining Australia as 'first launch country', meaning that Australia should be at the front of the queue for access to new medicines.
As one of the people who was at the table when various PBS reforms were negotiated with government 20 years ago, I fear that PBS and HTA reform is at risk of being labelled a failure.
It genuinely pains me to say this. I'm invested in it and I see it as a chance to make good public policy for the good of the Australian community and economy.
I fear that over the last two decades PBS reform has morphed from a considered policy reform agenda into a simple revenue raising exercise. A grab for cash.
A positive way forward: the PBS as a productivity driver
The good news is that there is real opportunity for Australia now.
There is the potential to capitalise on the current circumstances and start delivering on the promise of PBS reform and make up for 20 years of lopsided policy drift.
The pharmaceutical industry has worked well with governments - Labor and Coalition - for the last two decades to provide the financial headroom for new innovation. Through groups like Medicines Australia, the industry has done a great job of being part of the policy solution for governments. This is important, ground-breaking work that deserves recognition.
The current Minister for Health, Mark Butler, has reports on his desk on how to start reforming the HTA system so that Australia's health system can adopt a swathe of new, emerging technologies in healthcare. Not just innovative medicines, but innovative vaccines, devices, genomic technologies and digital technologies to lead the Australian health system into the 21st century.
There is more and more evidence emerging about the role of innovative new medicines in driving productivity growth in Australia's health system and broader economy. At a time when Australia's productivity growth performance risks future economic prosperity, there's now hard evidence that new medicines and other technologies and interventions in the health system are key tools to improve Australia's productivity performance.
Unfortunately, the current HTA framework used by the Australian Government doesn't recognise this. While things might be changing, historically the Department of Health, the PBAC, and economic departments in government either haven't understood or haven't valued these productivity benefits.
This has to change. It's time to bring our HTA frameworks into the 21st century.
Federal governments, the pharmaceutical industry, and patient groups have spent the last 20 years shoring up the financial sustainability of the PBS and providing the financial headroom for new innovation. We should spend the next 20 years delivering on the benefits of that for Australian patients, the community and the broader economy.
So, here's a suggested four-point plan for the Australian Government to get this started:
Update Australia's HTA assumptions and methods for the 21st century and develop a plan to go beyond the recommendations of the HTA Review
Recognise that industry has already made billions of dollars in down payments for reform improvements through PBS savings measures
Recognise the evidence of the economic and productivity value of new medical technologies, and
Invest more in the capital and technology in healthcare to drive productivity and patient outcomes.
That would be a promise worth waiting for.





Comments